Summary
Regeneron has received FDA approval for Otarmeni (lunsotogene parvec-cwha), the first gene therapy for genetic hearing loss caused by biallelic OTOF variants, marking a major milestone in sensory-organ gene therapy.
What Happened
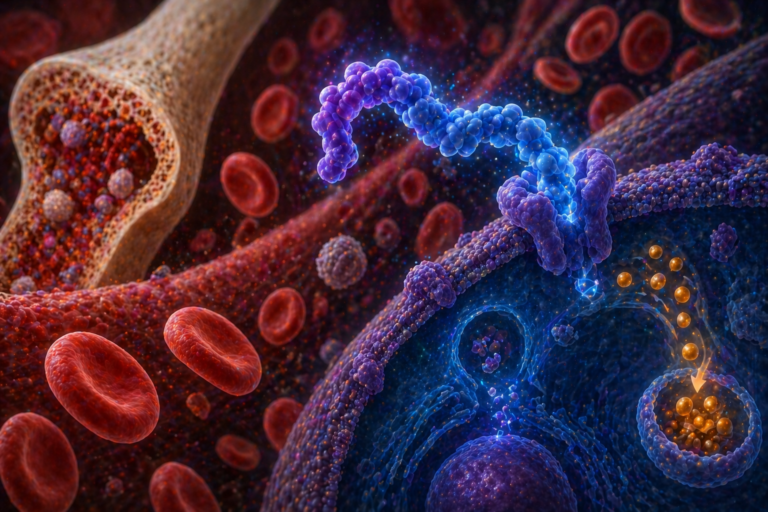
The FDA approved Otarmeni for patients with ultra-rare genetic hearing loss driven by OTOF mutations. The therapy is delivered via an AAV-based gene therapy approach designed to restore functional otoferlin protein in inner ear cells. The approval also leverages the FDA’s priority voucher framework for rare conditions.
Deep Analysis
This approval represents a significant platform validation for gene therapy beyond traditional targets such as liver, eye, and hematologic disorders. The inner ear has long been considered a challenging environment for gene delivery due to accessibility and cellular specificity requirements.
Successful approval suggests that targeted delivery to sensory organs is becoming clinically viable. This expands the addressable landscape for gene therapy into neurosensory diseases.
Strategically, this positions Regeneron at the forefront of next-generation gene therapy applications and reinforces continued momentum in AAV-based approaches. It also signals that ultra-rare disease pathways can serve as entry points for broader platform validation.
From a competitive standpoint, this may catalyze increased investment in sensory-organ gene therapies, including ophthalmology, neurology, and hearing-related indications.
Company / Product Background
Regeneron is a leading biopharmaceutical company with strengths in biologics and emerging gene therapy platforms.
Genetic hearing loss caused by OTOF mutations results from absence of functional otoferlin protein, which is critical for synaptic transmission in auditory hair cells. This leads to impaired signal transmission from the inner ear to the brain.
Otarmeni is an AAV-based gene therapy designed to deliver a functional copy of the OTOF gene directly to inner ear cells, restoring protein expression and enabling proper auditory signaling.
Signal Extraction
– First approval in genetic hearing loss gene therapy
– Validation of AAV delivery in sensory organs
– Expansion of gene therapy beyond traditional tissues
– Rare disease pathway enabling platform proof
Insilens Take
– Opportunity: Expansion into sensory-organ gene therapy platforms
– Threat: Technical complexity of localized delivery
– Watch Signal: Durability and real-world efficacy data
– Action: Track next targets in neurosensory gene therapy