Summary
The RNA therapeutics field is approaching a key inflection point with the anticipated initiation of the first clinical trial of engineered transfer RNA (tRNA) therapy, led by emerging players such as Alltrna. This modality targets nonsense mutations, representing a new class of genetic medicine beyond mRNA, siRNA, and antisense oligonucleotides.
What Happened
Preclinical development of engineered tRNA therapies has progressed to the stage where first-in-human clinical trials are expected to begin. These therapies are designed to restore protein translation in diseases caused by nonsense mutations by enabling ribosomes to read through premature stop codons.
Initial development is focused on liver-targeted genetic diseases, where delivery and expression are more tractable, with potential expansion to broader indications over time.
Deep Analysis
This development represents a true category-creation moment in RNA therapeutics. While mRNA, siRNA, and antisense approaches have validated RNA as a therapeutic modality, tRNA therapy introduces a fundamentally different mechanism—directly intervening in the translation process.
Nonsense mutations account for a significant proportion of genetic diseases, making this approach potentially broad in scope. Unlike gene replacement or editing strategies, tRNA therapy could provide a repeatable, modular solution across multiple diseases sharing similar mutation types.
However, the modality remains unproven in humans. Key risks include delivery efficiency, specificity of codon recognition, off-target translation effects, and durability of response. Early clinical data will be critical in determining whether this platform can achieve meaningful therapeutic windows.
From a strategic standpoint, if successful, tRNA therapies could evolve into a platform comparable to early RNA therapeutics or CRISPR, with wide-ranging applications across rare disease, oncology, and potentially hematologic disorders.
Company / Product Background
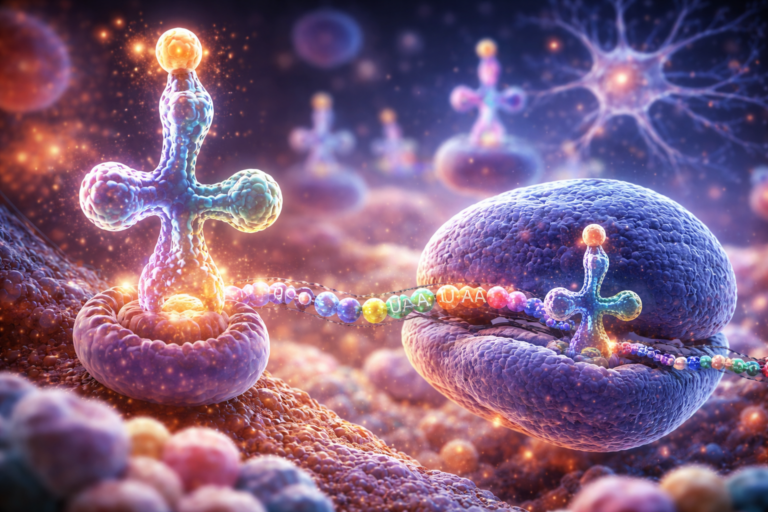
Alltrna is a biotechnology company developing engineered tRNA therapies to treat genetic diseases caused by nonsense mutations. The platform focuses on designing synthetic tRNAs that can insert amino acids at premature stop codons, enabling production of full-length functional proteins.
Genetic diseases caused by nonsense mutations result from premature stop codons that truncate protein synthesis, leading to loss of function. These mutations are implicated in a wide range of rare diseases, particularly those affecting the liver and metabolic pathways.
Engineered tRNA therapies work by delivering modified tRNA molecules that recognize stop codons and allow ribosomes to continue translation, restoring protein production. This approach aims to correct the underlying defect at the level of protein synthesis rather than altering DNA or RNA transcription.
Signal Extraction
– Emergence of a new RNA therapeutic class beyond existing modalities
– Platform potential across multiple genetic diseases driven by nonsense mutations
– Early-stage, high-risk but high-optionality innovation signal
– Could follow trajectory of mRNA or CRISPR if clinically validated
Insilens Take
– Opportunity: Early identification of tRNA platform leaders and partnership targets
– Threat: High technical risk and uncertain clinical translation
– Watch Signal: First human data demonstrating safety and functional protein restoration
– Action: Track modality development and compare to adjacent RNA approaches
Importance & Confidence
Importance: High
Confidence: Medium